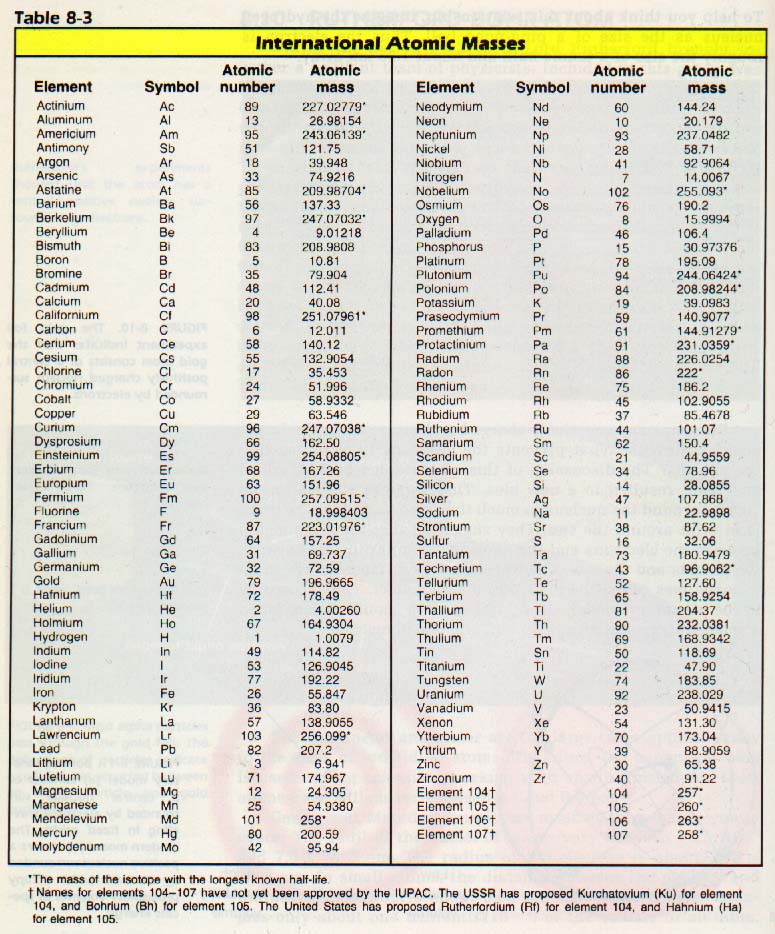
Periodic Table: Unique 4-letter Combinations, Part IV. To confirm your answer, compare the calculated value to the weighted mass displayed on the periodic table. Molar masses of Elements Can you name the Molar masses of Elements By chemistrymaster. The atomic mass of oxygen (use percent natural abundance data from Table 2.5.1) would be calculated as follows:Ītomic mass = (%1)(mass 1) + (%2)(mass 2) + (%3)(mass 3) For chlorine, there are only two naturally occurring isotopes so there are only two terms.Ītomic mass = (%1)(mass 1) + (%2)(mass 2) + ⋯Īnother example: oxygen exists as a mixture that is 99.759% 16O, 0.037% 17O and 0.204% 18O. This is repeated until there is a term for each isotope. The weighted average is determined by multiplying the percent of natural abundance by the actual mass of the isotope. The atomic mass of an element is the weighted average of the atomic masses of the naturally occurring isotopes of that element. Why? The reason is that we need to take into account the natural abundance percentages of each isotope in order to calculate what is called the weighted average.

If the mass of a substance is known, the number of moles in the substance can be calculated. The atomic masses of some elements are known very precisely, to a large number of decimal places. The molar mass of an element is found on the periodic table, and it is the elements atomic weight in grams/mole (g/mol). The periodic table provides the atomic mass next to or below each element. We have defined a mole based on the isotopic atomic mass of carbon-12. The substances mass is the sum of all of those atomic masses. The atomic mass of an element is the relative average of all of the naturally occurring isotopes of that element and atomic mass is the number that appears in the periodic table. You can click and download it from below.= 35.968 \,u \nonumber \]Īs you can see, the average atomic mass given in the last column of the table above (35.453) is significantly lower. The atomic masses in Table A.1 The Basics of the Elements of the Periodic Table represent the number of decimal places recognized by the International Union of Pure and Applied Chemistry, the worldwide body that develops standards for chemistry. The elements that compose a substance all have an atomic mass. If you want to know about What is the modern p table of elements? And are looking for a periodic table of elements, you are in the right place because we are providing PDF. Therefore the energy level of an atom increases, the number of sub-level energy level also increases. Electron’s number in a period increases as one moves down the periodic table. The rows in the table are called periods.Įlement’s periodic number signifies the highest energy level an electron occupies. Elements are arranged from top to bottom and from left to right in increasing order of their increasing atomic number and coincide with increasing atomic mass. The periodic table of elements put the entire chemical element in order. Scientific calculator for chemists - can be used as a common scientific calculator (sin, cos, log, power, root, memory), but also contains a molecular weight calculator and tables with various physical and chemical constants. Rare earth elements (REE) - Rare earth elements (REE) are a collection of seventeen chemical elements in the periodic table, specifically the fifteen lanthanides plus scandium and yttrium.
The element names are as large as I could consistently make them. This periodic table includes all 118 elements known as of 2017. Buy print or eBook Opens in a new window Book contents. Calculator - the best online scientific calculator. This color periodic table contains the usual element numbers, symbols, element names, and atomic masses, but has the added bonus of larger type. How Are The Elements Arranged In The Modern p Table Periodic table of the elements, with molar masses A Life Scientists Guide to Physical Chemistry. He arranged the elements in the increasing order of their atomic number.
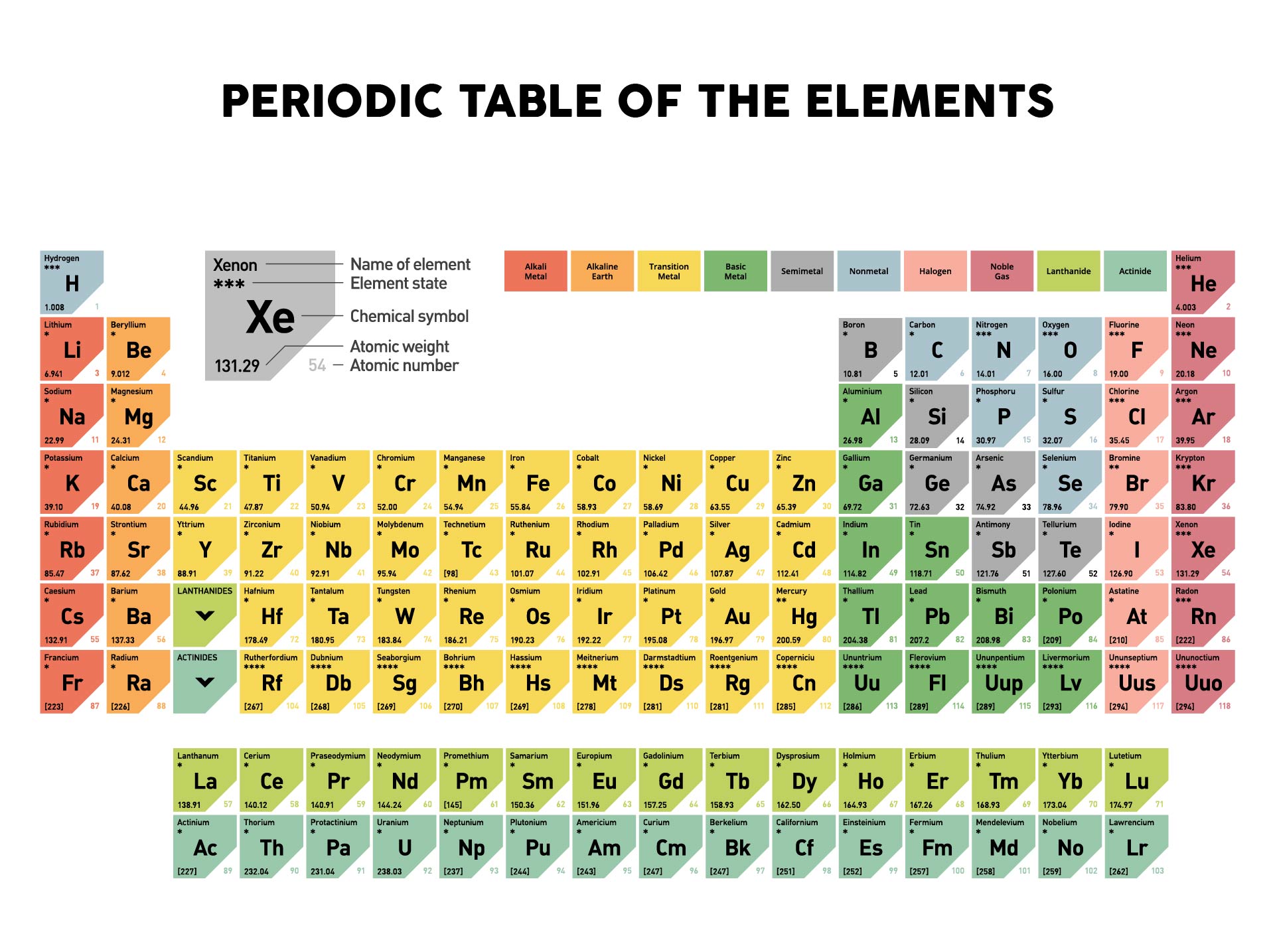
He arranged all the chemical elements in order of their atomic mass corresponding to their relative molar mass. Dmitri Mendeleev, a Russian chemist has given the first periodic table.
